Blog
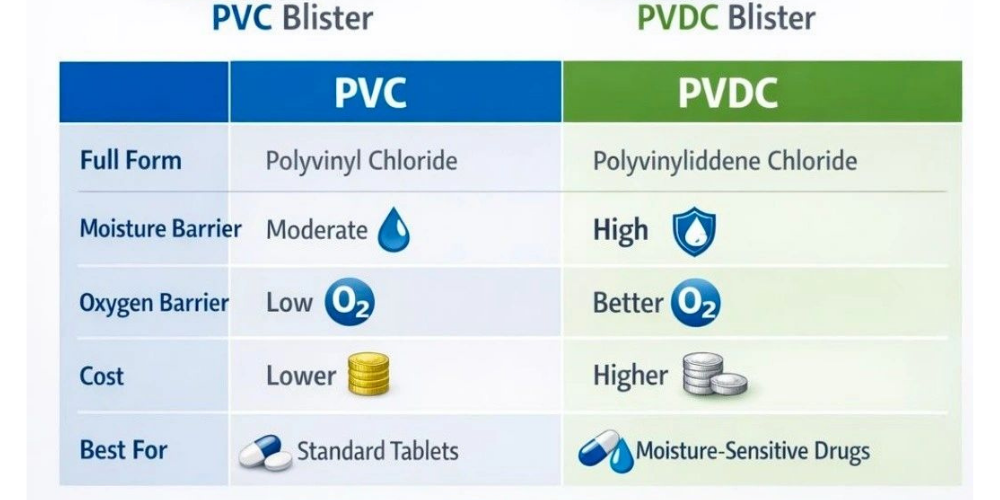
Difference Between PVC and PVdC Blister Packaging
One common question in solid oral dosage packaging is the difference between PVC and PVDC blister materials. PVC (Polyvinyl Chloride) ...

How Do You Choose an RPM in Dissolution?
I’ve seen a couple of “A.I. Slop” articles on LinkedIn this week about how to choose an RPM speed in ...

How to Identify Critical Material Attributes (CMAs) of an API in QbD
In QbD, we often focus heavily on formulation and process variables. But a large part of product performance actually depends ...

Extent of CMA Evaluation Required for Non-Critical Excipients
In development, especially within a QbD framework, there is frequent debate on whether non-critical excipients need detailed characterization of their ...

Statistics Required in Formulation Development
1. Formulation development is not trial and error. 2. The purpose of statistics in pharmaceutical R and D is not ...

A Bioequivalence Lesson Experience
As R&D leaders, we often approve programs that look flawless on paper. Same API. Same grade. Formulation physicochemical characteristics aligned. ...

Do Pharmacopeial Methods Need Forced Degradation Studies?
It’s a fairly common question. Usually built on assumptions like: So, the conclusion becomes: 𝘞𝘩𝘺 𝘋𝘖 𝘧𝘰𝘳𝘤𝘦𝘥 𝘥𝘦𝘨𝘳𝘢𝘥𝘢𝘵𝘪𝘰𝘯 𝘢𝘵 𝘢𝘭𝘭? ...

Does f2 Apply to Modified-Release Dissolution Profiles?
Short answer: Yes. But only if you meet the prerequisites Most of us learn f2 through immediate-release examples. That’s why ...

Root Cause Analysis in Pharmaceutical Industry
Root cause analysis (RCA) is a systematic process used in the pharmaceutical industry to identify the underlying factors that contribute ...

Bound Water vs Free Water
Understanding water states is essential in formulation, stability, and lyophilization. Here’s the simplest breakdown: Bound Water Free Water Why it ...

Computer System Assurance
Computer System Assurance (CSA) refers to the comprehensive and systematic approach taken by organizations to ensure the reliability, security, and ...

CSV Qualification Summary Report
A Computer System Validation (CSV) Qualification Summary Report is a comprehensive document that provides an overview of the validation process ...
