Chief Pharmacist

Clean Out of Place (COP)
Equipment is cleaned out of place as a whole (i.e., tanks) or as a part disassembled from a larger system. ...

Manual Cleaning Process
Manual cleaning processes continue to play a role in the pharmaceutical industry. Some equipment and parts are not easily adapted ...

Clean in Place (CIP) in Pharmaceutical Industry
Principles of CIP The underlying principle of CIP is to remove undesirable actives and chemicals from product contact surfaces by ...

CIP Cycle Development
Cleaning cycles vary by industry; however, they are based on the same principles of relying on TACT. Various water- soluble ...

How Pharmacogenomics Benefits Patients: Real-World Examples
Tamoxifen in breast cancer patients is one of the best examples illustrating how pharmacogenomics can provide drug dosing guidance. Tamoxifen ...

How Does Genotype Influence Drug Metabolism?
Like all DNA, the genes that encode drug‑metabolizing enzymes (DMEs) and other proteins that interact with drugs are subject to ...

Pharmacogenomics: The Foundation of Personalized Therapy
Although modern medical science has developed an extensive pharmacopeia to target a wide variety of pathologies, we know that not ...

Common Definitions in Pharmacogenomics
Allele One of a number of alternative forms of the same gene or the same genetic locus. Allele specific SNP ...

Dissolution Vs. Disintegration: What is Right for your Product?
Dissolution and disintegration are both commonly used for checking the quality of solid oral dosage forms. While these two techniques ...

Dissolution and Dissolved Gasses: How Should You Handle Them?
Dissolved gasses in your dissolution media can cause a variety of issues to your run. If not controlled, these gasses ...

Key Parts of DMF to Review
When developing generic products, reviewing the Drug Master File (DMF) is a critical step. Finished product manufacturers are expected to ...
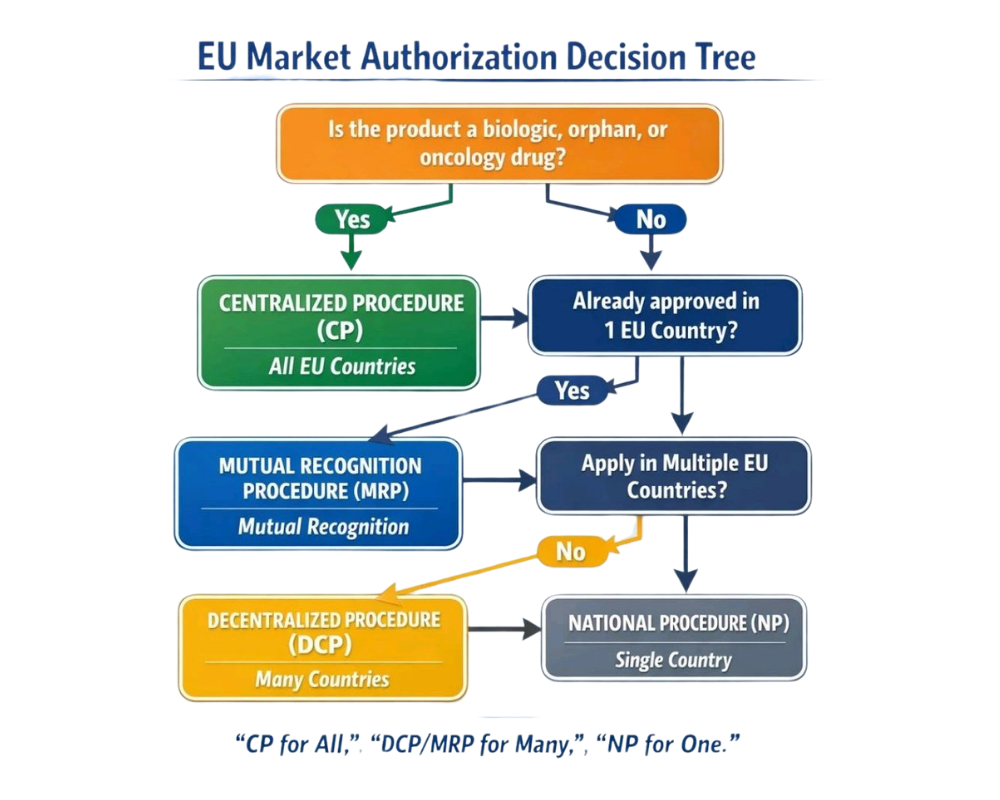
EU Market Authorization: A Strategic Decision Tree for Pharma Professionals
Navigating the EU regulatory landscape is critical for successful product launches. Choosing the right authorization pathway is not just procedural, ...
